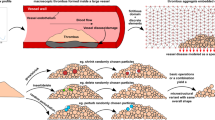
Abstract
The mouse laser injury thrombosis model provides up to 0.22 μm-resolved voxel information about the pore architecture of the dense inner core and loose outer shell regions of an in vivo arterial thrombus. Computational studies were conducted on this 3D structure to quantify transport within and around the clot: Lattice Boltzmann method defined vessel hemodynamics, while passive Lagrangian Scalar Tracking with Brownian motion contribution simulated diffusive-convective transport of various inert solutes (released from lumen or the injured wall). For an input average lumen blood velocity of 0.478 cm/s (measured by Doppler velocimetry), a 0.2 mm/s mean flow rate was obtained within the thrombus structure, most of which occurred in the 100-fold more permeable outer shell region (calculated permeability of the inner core was 10−11 cm2). Average wall shear stresses were 80–100 dyne/cm2 (peak values >200 dyne/cm2) on the outer rough surface of the thrombus. Within the thrombus, small molecule tracers (0.1 kDa) experienced ~70,000 collisions/s and penetrated/exited it in about 1 s, whereas proteins (~50 kDa) had ~9000 collisions/s and required about 10 s (tortuosity ~2–2.5). These simulations help define physical processes during thrombosis and constraints for drug delivery to the thrombus.








Similar content being viewed by others
References
Agard, D. A., Y. Hiraoka, P. Shaw, and J. W. Sedat. Fluorescence microscopy in 3 dimensions. Methods Cell Biol. 30:353–377, 1989.
Barber, C. B., D. P. Dobkin, and H. Huhdanpaa. The Quickhull algorithm for convex hulls. ACM Trans. Math. Softw. 22(4):469–483, 1996.
Bellido-Martin, L., V. Chen, R. Jasuja, B. Furie, and B. C. Furie. Imaging fibrin formation and platelet and endothelial cell activation in vivo. Thromb. Haemost. 105(5):776–782, 2011.
Bhatnagar, P. L., E. P. Gross, and M. Krook. A model for collision processes in gases. 1. Small amplitude processes in charged and neutral one-component systems. Phys. Rev. 94(3):511–525, 1954.
Borders, J. L., and H. J. Granger. An optical Doppler intravital velocimeter. Microvasc. Res. 27(1):117–127, 1984.
Boyd, J., J. Buick, and S. Green. A second-order accurate lattice Boltzmann non-Newtonian flow model. J. Phys. A 39(46):14241–14247, 2006.
Brass, L. F., K. M. Wannemacher, P. Ma, and T. J. Stalker. Regulating thrombus growth and stability to achieve an optimal response to injury. J. Thromb. Haemost. 9(s1):66–75, 2011.
Brinkman, H. C. A calculation of the viscous force exerted by a flowing fluid on a dense swarm of particles. Appl. Sci. Res. 1(1):27–34, 1947.
Chen, S., and G. D. Doolen. Lattice Boltzmann method for fluid flows. Annu. Rev. Fluid Mech. 30:329–364, 1998.
Colace, T., E. Falls, X. L. Zheng, and S. L. Diamond. Analysis of morphology of platelet aggregates formed on collagen under laminar blood flow. Ann. Biomed. Eng. 39(2):922–929, 2011.
Comerford, A., M. J. Plank, and T. David. Endothelial nitric oxide synthase and calcium production in arterial geometries: an integrated fluid mechanics/cell model. J. Biomech. Eng. Trans. ASME 130(1):011010, 2008.
Cosgrove, J. A., J. M. Buick, S. J. Tonge, C. G. Munro, C. A. Greated, and D. M. Campbell. Application of the Lattice Boltzmann method to transition in oscillatory channel flow. J. Phys. A 36(10):2609–2620, 2003.
Davis, M. J. Determination of volumetric flow in capillary tubes using an optical Doppler-velocimeter. Microvasc. Res. 34(2):223–230, 1987.
Desjardins, C., and B. R. Duling. Microvessel hematocrit—measurement and implications for capillary oxygen-transport. Am. J. Physiol. 252(3):H494–H503, 1987.
Einstein, A. The motion of elements suspended in static liquids as claimed in the molecular kinetic theory of heat. Ann. Phys. 17(8):549–560, 1905.
Falati, S., P. Gross, G. Merrill-Skoloff, B. C. Furie, and B. Furie. Real-time in vivo imaging of platelets, tissue factor and fibrin during arterial thrombus formation in the mouse. Nat. Med. 8(10):1175–1181, 2002.
Gabbanelli, S., G. Drazer, and J. Koplik. Lattice Boltzmann method for non-Newtonian (power-law) fluids. Phys. Rev. E 72(4):046312–046317, 2005.
Granger, D. N., J. N. Benoit, M. Suzuki, and M. B. Grisham. Leukocyte adherence to venular endothelium during ischemia-reperfusion. Am. J. Physiol. 257(5):G683–G688, 1989.
Holme, P. A., U. Orvim, M. J. A. G. Hamers, N. O. Solum, F. R. Brosstad, R. M. Barstad, and K. S. Sakariassen. Shear-induced platelet activation and platelet microparticle formation at blood flow conditions as in arteries with a severe stenosis. Arterioscler. Thromb. Vasc. Biol. 17(4):646–653, 1997.
Kandhai, D., A. Koponen, A. G. Hoekstra, M. Kataja, J. Timonen, and P. M. A. Sloot. Lattice-Boltzmann hydrodynamics on parallel systems. Comput. Phys. Commun. 111(1–3):14–26, 1998.
Kim, M. B., and I. H. Sarelius. Distributions of wall shear stress in venular convergences of mouse cremaster muscle. Microcirculation 10(2):167–178, 2003.
Levine, H. A., M. P. McGee, and S. Serna. Diffusion and reaction in the cell glycocalyx and the extracellular matrix. J. Math. Biol. 60(1):1–26, 2010.
Lipowsky, H. H. Microvascular rheology and hemodynamics. Microcirculation 12(1):5–15, 2005.
Lipowsky, H. H., S. Kovalcheck, and B. W. Zweifach. Distribution of blood rheological parameters in microvasculature of cat mesentery. Circ. Res. 43(5):738–749, 1978.
Matsumoto, M., and T. Nishimura. Mersenne twister: A 623-dimensionally equidistributed uniform pseudo-random number generator. ACM Trans. Model. Comput. Simul. (TOMACS) 8(1):3–30, 1998.
Merrill, E. W., and G. A. Pelletie. Viscosity of human blood—transition from Newtonian to non-Newtonian. J. Appl. Physiol. 23(2):178–182, 1967.
Nesbitt, W. S., E. Westein, F. J. Tovar-Lopez, E. Tolouei, A. Mitchell, J. Fu, J. Carberry, A. Fouras, and S. P. Jackson. A shear gradient-dependent platelet aggregation mechanism drives thrombus formation. Nat. Med. 15(6):665–673, 2009.
Papavassiliou, D. V. Turbulent transport from continuous sources at the wall of a channel. Int. J. Heat Mass Transf. 45(17):3571–3583, 2002.
Papavassiliou, D. V., and T. J. Hanratty. The use of Lagrangian-methods to describe turbulent transport of heat from a wall. Ind. Eng. Chem. Res. 34(10):3359–3367, 1995.
Porter, B., R. Zauel, H. Stockman, R. Guldberg, and D. Fyhrie. 3-D computational modeling of media flow through scaffolds in a perfusion bioreactor. J. Biomech. 38(3):543–549, 2005.
Qian, Y. H., D. Dhumieres, and P. Lallemand. Lattice BGK models for Navier–Stokes equation. Europhys. Lett. 17(6):479–484, 1992.
Sakariassen, K. S., P. A. Holme, U. Orvim, R. M. Barstad, N. O. Solum, and F. R. Brosstad. Shear-induced platelet activation and platelet microparticle formation in native human blood. Thromb. Res. 92(6):S33–S41, 1998.
Skartsis, L., J. L. Kardos, and B. Khomami. Resin flow through fiber beds during composite manufacturing processes. 1. Review of Newtonian flow through fiber beds. Polym. Eng. Sci. 32(4):221–230, 1992.
Stalker, T. J., E. A. Traxler, J. Wu, K. M. Wannemacher, S. L. Cermignano, R. S. Voronov, S. L. Diamond, and L. F. Brass. Hierarchical organization in the hemostatic response and its relationship to the platelet signaling network. Blood (blood-2012-09-457739; published ahead of print January 9, 2013, doi:10.1182/blood-2012-09-457739), 2012.
Succi, S. The Lattice Boltzmann equation for fluid dynamics and beyond. New York: Clarendon Press/Oxford University Press, 2001.
Sukop, M. C., D. T. Thorne, and NetLibrary Inc. Lattice Boltzmann Modeling an Introduction for Geoscientists and Engineers. Berlin: Springer, ix, 172 pp., 2006.
Swift, M. R., E. Orlandini, W. R. Osborn, and J. M. Yeomans. Lattice Boltzmann simulations of liquid–gas and binary fluid systems. Phys. Rev. E 54(5):5041–5052, 1996.
VanGordon, S., R. S. Voronov, T. B. Blue, R. L. Shambaugh, D. V. Papavassiliou, and V. I. Sikavitsas. Effects of scaffold architecture on preosteoblastic cultures under continuous fluid shear. Ind. Eng. Chem. Res. 50(2):620–629, 2010.
Wang, J. Y., X. X. Zhang, A. G. Bengough, and J. W. Crawford. Domain-decomposition method for parallel Lattice Boltzmann simulation of incompressible flow in porous media. Phys. Rev. E 72(1):016706–016711, 2005.
Wang, W. W., and M. R. King. Multiscale modeling of platelet adhesion and thrombus growth. Ann. Biomed. Eng. 40(11):2345–2354, 2012.
Windberger, U., A. Bartholovitsch, R. Plasenzotti, K. J. Korak, and G. Heinze. Whole blood viscosity, plasma viscosity and erythrocyte aggregation in nine mammalian species: reference values and comparison of data. Exp. Physiol. 88(3):431–440, 2003.
Xu, Z. L., N. Chen, S. C. Shadden, J. E. Marsden, M. M. Kamocka, E. D. Rosen, and M. Alber. Study of blood flow impact on growth of thrombi using a multiscale model. Soft Matter 5(4):769–779, 2009.
Yoshino, A., Y. Hotta, T. Hirozane, and M. Endo. A numerical method for incompressible non-newtonian fluid flows based on the Lattice Boltzmann method. J. Nonnewton. Fluid Mech. 147(1–2):69–78, 2007.
Acknowledgments
We acknowledge financial support from NIH R01-HL103419 and AHA 11POST6890012 grants. Computation was carried under TERAGRID supercomputing allocation TG-IBN110004 on the Lonestar linux cluster (Texas Advanced Computing Center). We would also like to acknowledge Dr. Papavassiliou’s Computational Transport Processes Laboratory at the University of Oklahoma, since a considerable portion of the code used in this study was written as a part of a PhD dissertation there. Finally, we would like to thank Dr. Henry J. Neeman, Dr. Raghu Reddy, and the whole OU Supercomputing Center for Education & Research (OSCER) team for useful discussions and suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Konstantinos Konstantopoulos oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Voronov, R.S., Stalker, T.J., Brass, L.F. et al. Simulation of Intrathrombus Fluid and Solute Transport Using In Vivo Clot Structures with Single Platelet Resolution. Ann Biomed Eng 41, 1297–1307 (2013). https://doi.org/10.1007/s10439-013-0764-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-013-0764-z