Abstract
BACKGROUND AND PURPOSE: Lesions revealed by pretreatment diffusion-weighted imaging (DWI) may not progress to infarction, and apparent diffusion coefficient (ADC) or DWI thresholds for tissue viability may exist. We evaluated the evolution of abnormal DWI findings in patients with acute stroke who underwent thrombolysis.
METHODS: Sixteen patients with acute occlusion of the anterior circulation underwent DWI followed by intra-arterial thrombolysis; follow-up CT or MR imaging was performed after reperfusion therapy. Lesion volumes were measured on all images. In three patients with abnormal DWI findings that appeared normal at follow-up, ADC values, ADC ratios, and DWI ratios were obtained on a section-by-section basis in the DWI-hyperintense regions that were either abnormal or normal at follow-up.
RESULTS: In three patients, part of the DWI-hyperintense tissue appeared normal and part appeared abnormal at follow-up imaging. In one of these patients, the lesion decreased by −35.6%. In the other two, lesion growth appeared in regions that appeared normal at initial DWI: in one, the overall change in size was −2.4%, and in the other, the lesion increased by 89.8%. Respective mean changes at follow-up in normal-appearing and abnormal-appearing regions were: ADC, 731.7 × 10−6 mm2/s and 650.4 × 10−6 mm 2/s; ADC ratio, 0.92 and 0.78; and DWI ratio, 1.16 and 1.32 (P < .001 for all measures).
CONCLUSION: In patients with acute stroke who undergo intra-arterial thrombolysis, most abnormal, pretreatment DWI findings indicate eventual infarction. In 19% of the patients described herein, DWI-hyperintense regions appeared normal at follow-up. ADC values, ADC ratios, and DWI ratios may be useful in identifying the portion of abnormal tissue that is potentially salvageable after reperfusion therapy.
Intra-arterial (IA) thrombolytics appear beneficial when given within 6 hours of stroke onset (1). To select the best candidates for thrombolysis, it is important to identify the characteristics of initial neuroimaging findings that may help predict tissue outcome. Selection criteria for thrombolysis are usually based on CT findings (1, 2). Patients with intracranial hemorrhage, CT findings of hypoattenuation throughout the affected vascular territory, or intracranial mass are typically excluded.
In the first hours after stroke onset, distinguishing ischemic tissue that is irreversibly injured from tissue that can be salvaged by reperfusion therapy is of great clinical value. Following vessel occlusion, there are two pathways for cell death: 1) cell necrosis that occurs in the acute and subacute stages and 2) apoptosis or delayed cell death that occurs during the chronic period. Diffusion-weighted imaging (DWI) has proved valuable for detecting the cell necrosis associated with acute ischemic brain tissue, with high sensitivity and specificity at early time points (3–5). In most patients imaged within 6 hours of stroke onset, the lesion grows from the time elapsed between initial DWI and follow-up CT or MR imaging, or the lesion size is unchanged (6–8). There are rare reports of spontaneous, substantial decreases in lesion size from the initial DWI abnormality to the follow-up infarct volume (5, 9, 10). Consequently, in the absence of thrombolysis, the DWI lesion is thought to represent a core of tissue that is destined to infarct.
IA thrombolysis has allowed higher recanalization rates at early time points, with improved clinical outcomes (1). In addition to recanalization grade, the time to recanalization, degree of collateralization, patient age, initial National Institutes of Health Stroke Scale (NIHSS) score, and clot location are correlated with clinical outcome scores and likely affect infarct growth (11–14). Animal experiments have demonstrated that, if the middle cerebral artery (MCA) is occluded for less than an hour or if the ADC values do not decrease below a certain threshold, diffusion lesion size decreases significantly (15–18). Furthermore, human studies have demonstrated substantial decreases in lesion size from the period between initial DWI and follow-up T2-weighted imaging in some patients receiving thrombolysis (19–21).
We hypothesized that, with thrombolysis, portions of the DWI abnormality may not progress to infarction and that there are ADC and DWI thresholds for tissue viability. The purposes of this study were 1) to determine how often abnormal tissue on DWI images does not appear to progress to infarction and 2) to compare DWI and ADC values in tissue that appears abnormal on initial DWI and follow-up images with tissue that appears abnormal on initial DWI but normal on follow-up images.
Methods
Patient Selection
Images of patients who underwent IA thrombolysis for the treatment of acute stroke between January 1, 2000, and April 30, 2001, were reviewed for this retrospective study. Inclusion criteria for subsequent analysis were the availability of DWI images obtained before IA thrombolysis and the availability of follow-up CT or T2-weighted MR images. Ten patients had undergone cranial CT as their only follow-up imaging study, and six patients underwent follow-up MR imaging. The study was approved by our institutional review board.
Imaging
MR imaging was performed on a 1.5-T whole-body system (Signa Echospeed; GE Medical Systems, Milwaukee, WI) with echo-planar capabilities. DWI images were obtained by using single-shot, echo-planar imaging with sampling of the entire diffusion tensor. Six high-b-value images corresponding to diffusion measurements in different gradient directions were acquired followed by a single low-b-value image. The high b value was 1000 s/mm2, and the low b value was 0 s/mm2. Imaging parameters were a TR/TE/NEX of 7500/100/3, an FOV of 22 × 22 cm, an image matrix of 128 × 128 pixels, a section thickness of 5 mm with a 1-mm gap, and 23 axial sections. Isotropic DWI images were generated offline on a network workstation (Sparcstation 20; Sun Microsystems, Milpitas, CA). ADC maps were also generated. This method has previously been described in detail (22, 23).
Fast spin-echo T2-weighted images were acquired with a TR/TEeff/NEX of 4200/102/1, an FOV of 20 cm, an acquisition matrix of 256 × 256 pixels, and a section thickness of 5 mm with a 1-mm gap.
Cranial CT was performed on a helical scanner (Advantage; GE Medical Systems) with 5-mm, contiguous, axial sections with 140 kVp, 340 mAs, and a 1-second scanning time.
Image Analysis
DWI images, calculated ADC maps, and follow-up CT or T2-weighted images were transferred to a personal computer for analysis. We used T2-weighted images, because we did not have fluid-attenuated inversion recovery images for all available follow-up MR studies. We did not use DWI images, because the ADCs in some tissue could be pseudonormalizing or returning to normal at follow-up. We compared the follow-up DWI and T2-weighted images and found no region that appeared abnormal on DWI but normal on T2-weighted images. Visually identified abnormalities on initial DWI and follow-up images were outlined by using a commercially available image display and analysis program (ALICE; Hayden Image Processing Solutions, Denver, CO). Two radiologists (P.W.S., A.H.) manually corrected the lesion contours on a section-by- section basis. Lesion volumes were calculated by summing individual section lesion areas multiplied by section separation (ie, section thickness plus intersection gap).
In 13 patients, all DWI hyperintense tissue appeared abnormal at follow-up. In three patients, part of the hyperintense tissue appeared normal, and part appeared abnormal at follow-up. In these three patients, the initial DWI images were coregistered with the follow-up CT or T2-weighted images with a commercially available coregistration program (Medex; Sensor Systems, Sterling, VA). Subsequently, two regions were defined on a section-by-section basis: Region 1 included abnormal areas on DWI images that appeared abnormal on follow-up CT or T2-weighted images. Region 2 included abnormal regions on DWI images that were normal on follow-up CT or T2-weighted images.
Areas similar to regions 1 and 2 were then identified in the normal contralateral brain. The mean DWI signal intensity from the abnormal side was divided by that of the contralateral normal side to obtain DWI ratios for each region. These regions were projected onto the ADC images. ADC ratios were similarly obtained for the two regions in each section and in each patient. Absolute ADC values were also obtained.
The extent of angiographic perfusion after treatment was classified according to thrombolysis in myocardial infarction (TIMI) grades (24).
Clinical Scoring
The NIHSS was used to assess initial neurologic status at admission, and the modified Rankin Scale (mRS) was used to assess neurologic status on the day of discharge. Both scores were obtained from the medical records.
Statistical Analysis
Two-tailed t tests were performed to compare mean lesion values in region 1 with mean lesion values in region 2 for ADC values, ADC ratios, and DWI ratios. Differences were considered statistically significant at P < .05.
Results
Demographic data and treatment characteristics are presented in Tables 1 and 2. The 16 patients were divided into two groups: group A, 13 patients without abnormal tissue on DWI images that appeared normal at follow-up, and group B, three patients with abnormal tissue on DWI images that appeared normal at follow-up (Fig 2). Mean patient age, mean initial NIHSS score, and mean time to treatment were similar for the two groups. Patients in group B all had MCA occlusions, whereas patients in Group A had occlusions of the MCA and internal carotid artery (ICA). All patients in group B were male, whereas 38% of patients in group A were male and 62% were female. Eleven patients (69%) had complete (two patients with TIMI grade 3) or partial (nine patients with TIMI grade 2) recanalization of thrombosed vessels at the conclusion of the procedure. The two patients with a TIMI grade of 3 had abnormal-appearing tissue at initial DWI that appeared normal at follow-up. In five patients (31%), there was no significant recanalization of thrombosed vessels (TIMI grade 1). One of these patients had abnormal-appearing tissue at DWI that appeared normal at follow-up. The mean mRS score at follow-up was approximately one point lower in group B than in group A.
Summary of demographic data and treatment characteristics
Details of demographic data and treatment characteristics
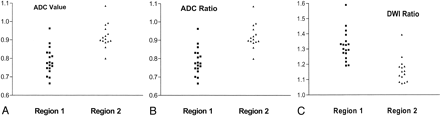
Of pixels initially hyperintense in all 16 patients, only 10.5% showed no abnormality at follow-up. Of initially hyperintense pixels in the three patients in group B, 41.0% showed no abnormality at follow-up. Figure 1 shows ADC values and ADC and DWI ratios for abnormal-appearing tissue at DWI that appeared abnormal at follow-up (region 1) and for abnormal-appearing tissue at DWI that appeared normal at follow-up (region 2) in the three patients in group B (Fig 2). The mean ADC value for region 2 was 731.7 × 10−6 mm2/s (range, 584.0–863.8 mm2/s; SD, 71.73), whereas the mean ADC value for region 1 was 650.4 × 10−6 mm2/s (range, 480.6–804.0 mm2/s; SD, 95.83). All abnormal-appearing regions on DWI images with ADCs less 584.0 × 10−6 mm2/s appeared abnormal at follow-up, and those with ADCs greater than 804.0 × 10−6 mm2/s appeared normal at follow-up. The mean ADC ratio for region 2 was 0.92 (range, 0.80–1.09; SD, 0.07), whereas the mean ADC ratio for region 1 was 0.78 (range, 0.67–0.96; SD, 0.07). All DWI-hyperintense regions with ADC ratios less than 0.80 appeared abnormal at follow-up, and those with ADC ratios greater than 0.96 appeared normal at follow-up. The mean DWI ratio for region 2 was 1.16 (range, 1.08–1.39; SD, 0.08), whereas, the mean DWI ratio for region 1 was 1.32 (range, 1.11–1.59; SD, 0.010). All DWI-hyperintense regions with DWI ratios greater than 1.39 appeared abnormal at follow-up, and those with DWI ratios less than 1.19 appeared normal at follow-up. Mean values for all parameters were significantly different between the two regions (P < .001).
Images obtained in three patients in group B who had hyperintense regions at initial DWI that appeared normal at follow-up. Region 1 is DWI-hyperintense tissue that was abnormal at follow-up imaging. Region 2 is DWI-hyperintense tissue that was normal at follow-up imaging.
A, ADC values in square millimeters per second.
B, ADC ratios.
C, DWI ratios.
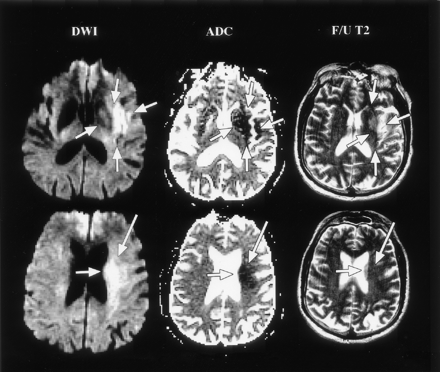
Reversal of DWI-hyperintense lesion after thrombolysis in a 68-year-old man with right-sided weakness and dysarthria. The patient had a left MCA occlusion that was successfully treated with IA tissue plasminogen activator at 6 hours. Initial DWI (row 1) and ADC (row 2) images demonstrate abnormality involving the left basal ganglia, insula, and subinsular region (short arrows) and in the left corona radiata (long arrow). At 3 days, follow-up (F/U) T2-weighted images demonstrate no abnormality in the left corona radiata. Lesions in the basal ganglia, insula, and subinsular region are unchanged. At discharge, the right-sided weakness had resolved.
Results of the volumetric analysis are presented in Table 3. Mean initial lesion volume in group A (65.75 cm3) was approximately twice that in group B (32.17 cm3). Also, the mean percentage increase in group A was 207.5% compared with 25.2% in group B. In one patient in group B, the lesion decreased from 37.3 to 24.0 cm3. Two patients in group B had regions with decreased diffusion that appeared normal at follow-up, as well as regions of initially normal tissue that appeared abnormal at follow-up: In one patient, the lesion marginally decreased from 16.8 to 16.4 cm3, and the other patient had an overall increase of 42.4 to 80.5 cm3. In all patients in group A, the lesion increased in size from the time of initial DWI to the follow-up CT or T2-weighted imaging. Patients without significant recanalization (TIMI grades 0 or 1) had a significantly larger mean percentage increase in infarct size (524.2%) compared with the change in those with partial or complete recanalization (114.4%, P < .05).
Volumetric data
Discussion
The existence of a threshold DWI or ADC value to indicate irreversible injury is controversial. We found that initial ADC values and DWI and ADC ratios were significantly different in abnormal-appearing tissue at DWI that appeared normal at follow-up imaging after IA thrombolysis in anterior-circulation strokes compared with those of regions remaining abnormal. Although there was overlap in values between the two regions, we were able to define outer limits for tissue that was likely to become infarcted even when treated with IA thrombolysis. All measured abnormal regions on DWI images with an ADC less than 584.0 × 10−6 mm2/s, an ADC ratio less than 0.80, or a DWI ratio greater than 1.39 appeared abnormal at follow-up. We also defined outer limits for DWI abnormal tissue that was likely to appear normal following thrombolysis. All measured DWI abnormal regions with an ADC greater than 804.0 × 10−6 mm2/s, an ADC ratio greater than 0.96, or a DWI ratio less than 1.19 appeared normal at follow-up. Further investigation is warranted to determine if these limits are useful in selecting patients who are most likely to benefit from thrombolysis.
Our results are in agreement with those from rat models of MCA occlusion suggesting ADC and DWI thresholds for tissue viability. Dardzinski et al (18) demonstrated a high correlation between tissue volume and ADC values below 550 × 10−6 mm2/s and between tissue volume and histologic infarction. Olah et al (25) demonstrated a high correlation between tissue volume and an ADC ratio of less than 0.77 and depletion of adenosine triphosphate below 30% that of the contralateral normal hemisphere. Hoehn-Berlage et al (26) demonstrated good agreement between tissue with an initial ADC ratio of <0.80 and histologic infarction. Minematsu et al (17) found a mean DWI ratio of 1.186 in DWI-hyperintense tissue that did not infarct and a mean ratio of 1.397 in tissue that did infarct.
Although numerous reports suggest the possibility of an ADC threshold, others indicate absence of a upper limit. Fiehler et al (27) demonstrated that final T2 lesion volume on day 7 was less than half the tissue volume with an initial ADC value of less than 60% of normal in two patients with successful early reperfusion. This is far below the threshold of 80% discussed before. They suggest that ADC normalization and tissue recovery depends on duration and severity of ischemia rather than the absolute ADC value. The concept of an increasing ADC threshold for tissue viability with increasing duration of ischemia should be considered in light of animal experiments that address perfusion thresholds for tissue viability. It has been shown that degree of ADC decrease is strongly associated with severity of perfusion deficits (28). Furthermore, Jones et al (29) demonstrated that cerebral blood flow threshold for tissue infarction increases with increasing occlusion time in a primate model. They found that the threshold was 10–12 mL/100 g/min for 2–3 hours of occlusion but 17–18 mL/100 g/min for permanent occlusion.
Judging whether tissue with a diffusion abnormality is normal at follow-up is complicated. Such lesions may appear normal on follow-up DWI, ADC, or T2-weighted images; however, this finding may not reflect complete tissue recovery, especially if the DWI images are obtained immediately following thrombolysis. Kidwell et al (30) reported a decrease in size of the initial abnormality disclosed by DWI performed immediately after IA thrombolysis in eight of 18 patients. However, despite the initial apparent recovery, a subsequent increase in DWI lesion volume was observed in five patients.
Animal studies have also demonstrated that tissue with DWI hyperintensity that appears normal on follow-up images may not reflect complete tissue recovery (31, 32). Rats undergoing transient MCA occlusion for 10 minutes, with DWI abnormality in the lateral caudoputamen, showed no abnormality on follow-up T2-weighted images at 72 hours. Histopathologic sections of the lateral caudoputamen demonstrated that 17% of neurons in this “salvaged” region were necrotic (31). Similarly, a study of 19 rats with MCA occlusion demonstrated mean decreases in lesion volume from the initial DWI to the follow-up T2-weighted imaging at 7 days of 25–60%, but the histologic infarctions were not significantly smaller than the initial DWI abnormalities (32). The authors proposed that the so-called MR fogging due to inflammatory infiltrate, gliosis, or petechial hemorrhage may account for the discrepancy.
Even with thrombolysis, the amount of DWI-hyperintense tissue that does not appear to infarct at follow-up imaging is relatively small. Kidwell et al (30) reported that eight of 18 patients who underwent successful IA thrombolysis had DWI-hyperintense regions that appeared normal at follow-up and that 41% of all DWI-revealed lesions did not progress to infarction. In our cohort, only 19% of our patients who underwent thrombolysis had DWI hyperintensities that were not persistently abnormal, and only 10.5% of all DWI hyperintensities appeared normal at follow-up. The lower percentages in our study may be related to the differences in treatment, time to treatment, recanalization rates, and time of follow-up imaging. The patients may also have differed in the severity of ischemia owing to different collateral flow patterns, cerebral perfusion pressure, and duration of hypoperfusion.
The primary limitation of our study was related to its design. Our study was retrospective, and routine clinical management, including decisions on the timing of imaging studies, were not altered for the purposes of this investigation. Therefore, times of initial and follow-up studies varied, and follow-up CT was performed in some cases because follow-up MR images were not available. MR images are more sensitive than CT scans in depicting tissue changes in acute and subacute stroke. Therefore, we may have overestimated extent of tissue that recovered and underestimated final infarct volume determined by follow-up CT compared with measures obtained by follow-up MR imaging. In addition, mean follow-up time of 3.2 days raises the question of whether some of the apparently reversible tissue might have demonstrated a secondary decline in ADC. If true, we are overestimating the amount of DWI-hyperintense tissue that may recover. Conversely, vasogenic edema could still be present at this time, and we could have overestimated the final infarct volume and underestimated the amount of abnormal-appearing tissue that might have recovered. Also, because apoptosis occurs in the chronic period, we did not consider this pathway for cell death in our calculations. For DWI and ADC value comparisons, we averaged regions rather than individual voxel values. It is unclear if these estimated thresholds help predict tissue viability in smaller regions. Although we assessed the infarct core, we did not analyze initially normal-appearing tissue on DWI images that progressed to infarction in the ischemic penumbra. Finally, there is error intrinsic to our coregistration program. The error for MR imaging-to-MR imaging registration is in the 75–150-μm range, whereas the median error for MR imaging-to-CT registration is in the 2–5-mm range (33, 34). This error likely caused misclassification of some pixels into region 1 versus region 2 and vice versa, and this error likely caused minor errors in volume calculations.
Conclusions
In most patients who undergo IA thrombolysis, we observe progression of a lesion to infarction in the interval between initial DWI and follow-up imaging. However, infarct growth is less in those with successful recanalization. In a minority of the patients described herein, we found that DWI-hyperintense regions could appear normal at follow-up. Furthermore, ADC values and ADC and DWI ratios were significantly different in DWI-hyperintense regions that progressed to infarction versus DWI-hyperintense regions that appeared normal at follow-up, although there was considerable overlap. Further investigation is warranted to determine whether there are DWI and ADC thresholds for tissue viability and to determine whether these are dependent on occlusion time, perfusion deficits, treatment, or a combination of numerous factors.
Acknowledgments
The authors wish to thank Cara O’Reilly for her help with image coregistration.
Footnotes
Supported in part by Public Health Service grant NS38477 (A.G.S.).
Presented in part at the Annual Meeting of the American Society of Neuroradiology, 2001.
References
- Received July 25, 2003.
- Accepted after revision December 12, 2003.
- Copyright © American Society of Neuroradiology